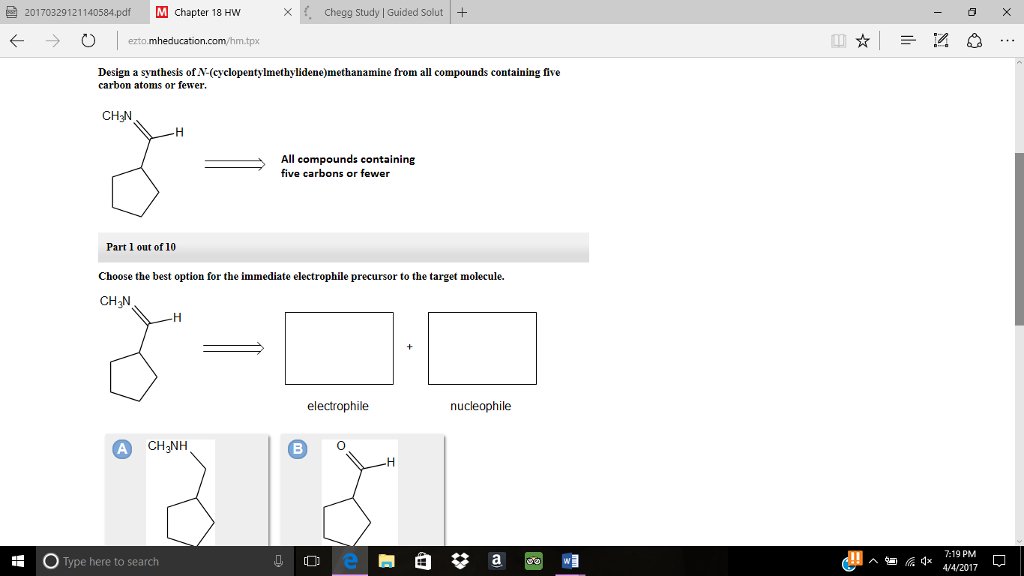 
Together, I translate heterolysis as a process in which a bond is broken and a pair of electrons is given to one atom, resulting in the other atom earning a positive charge. 
Hetero- means different or other, while -lysis means to break. To start, I compare heterolysis vs coordination. Break down the names of the reactions and explain the “tells” of the various steps.I particularly like flashcards because they help students with associative learning and with differentiating between the uses and types of bases. For example, topics like organometallics and hydride reagents need to be learned through practice (solving problems by hand) or other learning resources, such as flashcards. This is why I insist that my students constantly practice by putting their pens to paper, as discussed in an earlier post. Even though I hate saying this to my students, it’s important for them to know. Because some chemistry concepts don’t have any relevant analogies, they need to be learned through memorization. Sadly, there are some concepts that don’t have any real shortcuts, so students have to forgo the help of mnemonics or other tricks when learning the concepts and making solid connections. When I struggle to develop mnemonic devices or real-world examples for certain topics, I try to be honest with my students. /chapter3/pages17and18/page17and18_files/hidsigelec.png)
Keep in mind that the “e” and “a” in Lewis acids are meant to signify electrons and acceptors, respectively. ” On the other hand, L ewis acids, which are also electrophiles, are attacked. Some examples of nucleophiles include Lewis bases, and some examples of electrophiles include Lewis acids.Īnother way of looking at these two species-nucleophiles and electrophiles-is to remember that Lewis bases donate electrons because the “b” is a backwards “d. For instance, nucleophiles have a negative charge and need to give away their electrons, while electrophiles are positiv e and eat the electrons from the interacting reagents. Many times in the classroom, I use mnemonic devices to help my students recall the two species of nucleophiles and electrophiles. Always look at each reactionary step as the nucleophile vs the electrophile.With these notes in mind, I will share some of my best tactics for learning the elementary steps of organic chemistry: My notes are recorded in the Notability app from my iPad. To answer this question, I have included screenshots of the “Chapter Summary and Key Terms” from Chapter 7 below, which also contain my own suggestions to help my students tackle the challenging realm of organic reactions and mechanisms. In this year’s class, a student asked me, in a Perusall annotation, about the best way to learn this material. For those who are veterans, I hope to find your responses in the “Comments” section at the end! For those instructors who are still in their early years of academia, I write this post with you (and myself) in mind as we try to evolve in the classroom. With this in mind, I use each year to bolster my skills and presentation of the material. I have found that Chapter 7 (“An Overview of the Most Common Elementary Steps”), for example, is one of the harder chapters to teach. While some chapters are quite easy to present, others prove to be more challenging for students. However, mechanisms are not every student’s favorite topic. I am a big advocate of the Karty textbook and how it presents mechanisms to the audience.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
